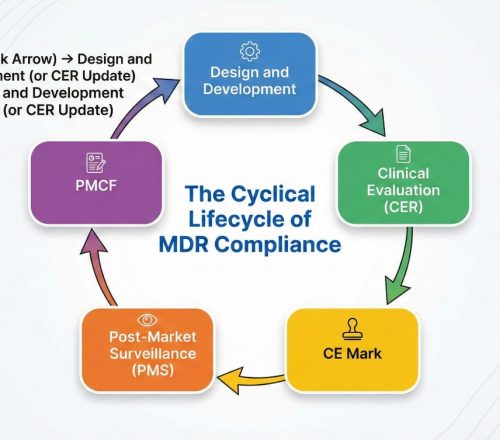
As MPH Clinical Research Center, we offer solutions that meet global standards not only for the clinical development of new drugs but also for life-saving medical devices. In addition to the comprehensive services we offer throughout all phases of drug development (Phase I-IV), we make a difference with our expertise in the CE Marking and EU Medical Device Regulation (MDR) compliance processes, which are the most critical needs of medical device manufacturers. By eliminating regulatory complexity, we manage your clinical data collection and reporting processes with speed, efficiency, and cost advantages, shortening your devices’ time to market. Thanks to our strategic location in Turkey and our international regulatory expertise, we provide the necessary clinical and regulatory strategies to confidently bring your products to the European market as well as rapidly growing Eastern markets such as China (NMPA), Thailand, and Korea.
Ongoing Studies
Loading..........
The Data is Not Available