The MDR Lifecycle Approach: The Critical Role of PMCF
The Medical Device Regulation (MDR) is based on the principle that the regulatory process does not end when a device receives the CE mark; rather, it transitions into a new phase: lifecycle management. One of the most critical components of this lifecycle is Post-Market Clinical Follow-up (PMCF).
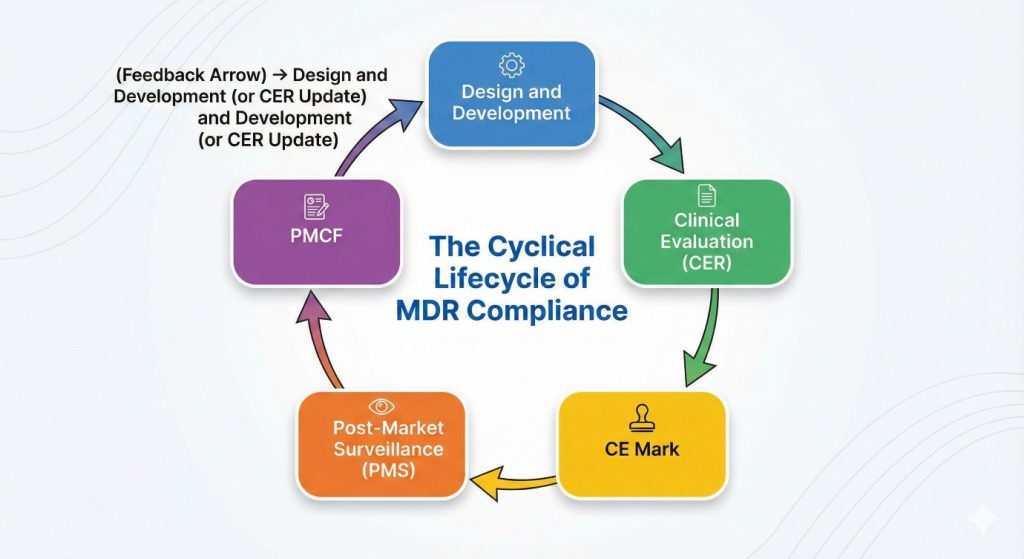
Figure 1: MDR Life Cycle (Cycle Diagram)
PMCF is the proactive process of collecting clinical data to continuously monitor the safety and performance of a device under its intended conditions of use after it has been placed on the market, and to keep the Clinical Evaluation Report (CER) up to date.
The Purpose of PMCF: Evidence from Real-World Data
The primary goal of PMCF is to address issues that could not be fully covered by clinical data collected before the device was placed on the market (Parts 5 and 6):
- Long-Term Safety and Performance: To identify the potential effects of long-term use and rare adverse events.
- Verification of Residual Risks: To confirm the acceptability of residual risks identified during the Risk Management process in a real-world setting.
- Early Detection of New Risks: To identify new risks or misuse that may emerge in the market at an early stage.
- CER Update: To continuously update the CER using the collected data and maintain the device’s conformity to the GSPRs (General Safety and Performance Requirements).
The PMCF Process: A Proactive and Planned Approach
PMCF is not a random data collection process but a systematic and planned approach.
- PMCF Plan
Every manufacturer must prepare a PMCF Plan appropriate for the risk class and characteristics of their device. This plan details what clinical data will be collected, by what methods (surveys, patient registries, new PMCF studies, etc.), and at what frequency.
- Data Collection and Analysis
Data is collected according to the plan. This data may include complaint records, literature reviews, user surveys, or specially designed PMCF studies (clinical investigations). The collected data is analyzed to determine if it affects the device’s benefit-risk ratio.
- PMCF Evaluation Report
The results of the analysis are summarized in a PMCF Evaluation Report at defined intervals (typically annually). This report forms the basis for updating the CER and is part of the Technical Documentation.
Continuous Compliance and Post-Market Surveillance (PMS)
PMCF is a component of the broader process known as Post-Market Surveillance (PMS). PMS encompasses all activities where the manufacturer proactively and reactively collects, records, and analyzes safety data throughout the device’s entire lifecycle.
Process | Scope | Focus |
PMS (Post-Market Surveillance) | All reactive and proactive activities (Complaint management, safety reporting, trend analysis). | General monitoring of the device’s safety in the market. |
PMCF (Post-Market Clinical Follow-up) | The proactive clinical data collection component of PMS. | Verification/update of the device’s clinical performance and the CER. |
MPH CTC’s Support for PMCF and Continuous Compliance
The MDR requires manufacturers to commit not just to a one-time CE approval but to continuous regulatory compliance. At MPH CTC, we help you manage this cyclical process:
- PMCF Plan Preparation: Medical support for creating cost-effective and MDR-compliant PMCF plans tailored to your device’s risk profile
- Execution of PMCF Studies: Conducting PMCF clinical investigations (e.g., long-term follow-up studies) under international GCP standards when necessary.
- CER Update Support: High-quality data support for regularly updating the CER in light of PMCF data and keeping the Technical Documentation ready for audit.
This continuous cycle of surveillance and data collection enhances your device’s market reliability and ensures success in Notified Body audits.
References
[1] Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices, Article 83.
[2] MDCG 2020-7 Guidance on PMCF Plan and Report.European Commission.
[3] The Role of PMCF in the Medical Device Lifecycle.NAMSA.