Introduction to the Concept of Medical Devices: The Backbone of Health Technologies
Medical devices, one of the cornerstones of modern healthcare, undertake the mission of protecting human health, diagnosing, treating, and improving quality of life. This broad spectrum, ranging from a simple bandage to AI-powered surgical robots, creates unique challenges for regulatory authorities. Therefore, clearly understanding whether a product is considered a “medical device” is critically important for both manufacturers and clinical researchers.
The European Union Medical Device Regulation (MDR) (EU) 2017/745 has clarified the definition of a medical device, establishing the regulatory framework in this field [1].
In-Depth Analysis of the MDR Definition
According to the MDR, a medical device is, essentially, any instrument, apparatus, appliance, software, implant, reagent, material, or other article intended by the manufacturer to be used for a specific medical purpose in humans.
“A medical device is:
1) Not intended to achieve its primary intended function by pharmacological, immunological, or metabolic action in or on the human body, but which may be assisted in achieving its function by such action, and specifically:
- i) Diagnosis, prevention, monitoring, prediction, prognosis, treatment, or alleviation of disease,
- ii) Diagnosis, monitoring, treatment, alleviation, or compensation for injury or disability,
iii) Investigation, replacement, or modification of anatomy or a physiological or pathological process or condition,
- iv) Provision of information through in vitro testing of specimens obtained from the human body, including organ, blood, and tissue donations,
devices, apparatus, equipment, software, implants, reagents, materials, or other substances designed by the manufacturer to be used alone or in combination on humans for one or more of these medical purposes.
2) Devices intended to prevent or support pregnancy,
3) Products specifically designed for the cleaning, disinfection, or sterilization of devices referred to in the second paragraph of Article 1 and devices referred to in subparagraph (1) of this paragraph [2]
Includes.
This definition sets out the three essential elements required for a product to be considered a medical device:
1 Intended Purpose: The medical purpose stated by the manufacturer on the device label, in the user manual, or in promotional materials.
2 Primary Mode of Action: The device does not perform its primary function through pharmacological, immunological, or metabolic means. This is the most critical point that distinguishes medical devices from drugs.
3 Comprehensive Category: It encompasses a wide range of materials and technologies, from physical hardware (devices, implants) to software (SaMD).
Key Difference Between Medical Devices and Medicines: Mode of Action
The most important factor determining a product’s regulatory pathway is its primary mode of action.
Feature | Medical Device | Medicine (Medical Product) |
Primary Mechanism of Action | Acts through physical, mechanical, thermal, optical, or software means. Functions through physical contact or interaction with the body. | Acts through pharmacological (chemical), immunological (immune system), or metabolic (biochemical processes) means. |
Regulation | MDR (Medical Device Regulation) and IVDR (In Vitro Diagnostic Regulation) | Medicinal Products Directive (Directive 2001/83/EC) |
Clinical Evidence | Clinical Evaluation Report (CER) and Clinical Investigations | Clinical Trials (Phase1-4) |
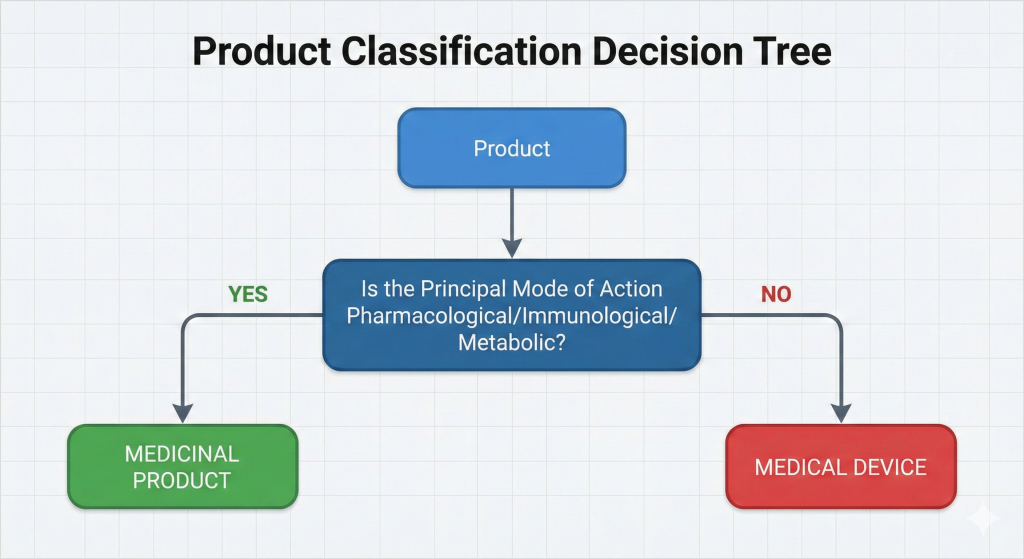
Figure 1: Product Classification Decision Tree
Intended Uses of Medical Devices
The medical purposes specified in the MDR definition demonstrate the wide range of applications for these devices. These purposes directly influence the classification of the device and consequently the approval process.
Intended Use | Description | Example Devices |
Diagnosis | To determine the presence, cause, or degree of a disease or condition. | MRI devices, X-ray devices, diagnostic software. |
Prevention | To prevent the onset of a disease or condition. | Special syringes used for vaccine administration, certain protective clothing. |
Monitoring | To track the course of a disease or physiological parameters. | Heart rate monitors, blood glucose meters. |
Treatment/Alleviation | To cure the disease or reduce its symptoms. | Surgical instruments, prostheses, pain relief devices. |
Anatomical/Physiological Process Change | To change or replace the body’s structure or function. | Pacemakers, artificial joints, stents. |
Types and Scope of Medical Devices
Medical devices are divided into various subcategories based on their physical form and function. The MDR has also included certain products that were previously unregulated.
- Active and Non-Active Devices
- Active Devices: Devices that require electrical energy or another power source to operate (e.g., ultrasound device, ventilator).
- Non-Active Devices: Devices that do not require any power source to operate (e.g., surgical sutures, bandages, catheters).
- Implants
Devices intended to be placed completely or partially in the body and remain there after the procedure is complete (e.g., heart valves, hip prostheses).
- Software as a Medical Device (SaMD)
One of the most significant innovations of the MDR is that it brings software that independently serves a medical purpose (e.g., mobile applications that analyze imaging data and provide diagnostic suggestions) under its regulatory scope.
Conclusion
The broad definition of medical devices and the strict rules introduced by the MDR require manufacturers to adopt a strategic approach to regulatory processes. In the next section, we will examine in detail how the risk-based classification of these devices (Class I, IIa, IIb, III) determines the approval process.
References
[1] Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.
[2] TIBBİ CİHAZ YÖNETMELİĞİ, Resmî Gazete Tarihi: 02.06.2021 Resmî Gazete Sayısı: 31499 Mükerrer