Part 2: EU Medical Device Regulation (MDR) and IVDR: The New Era
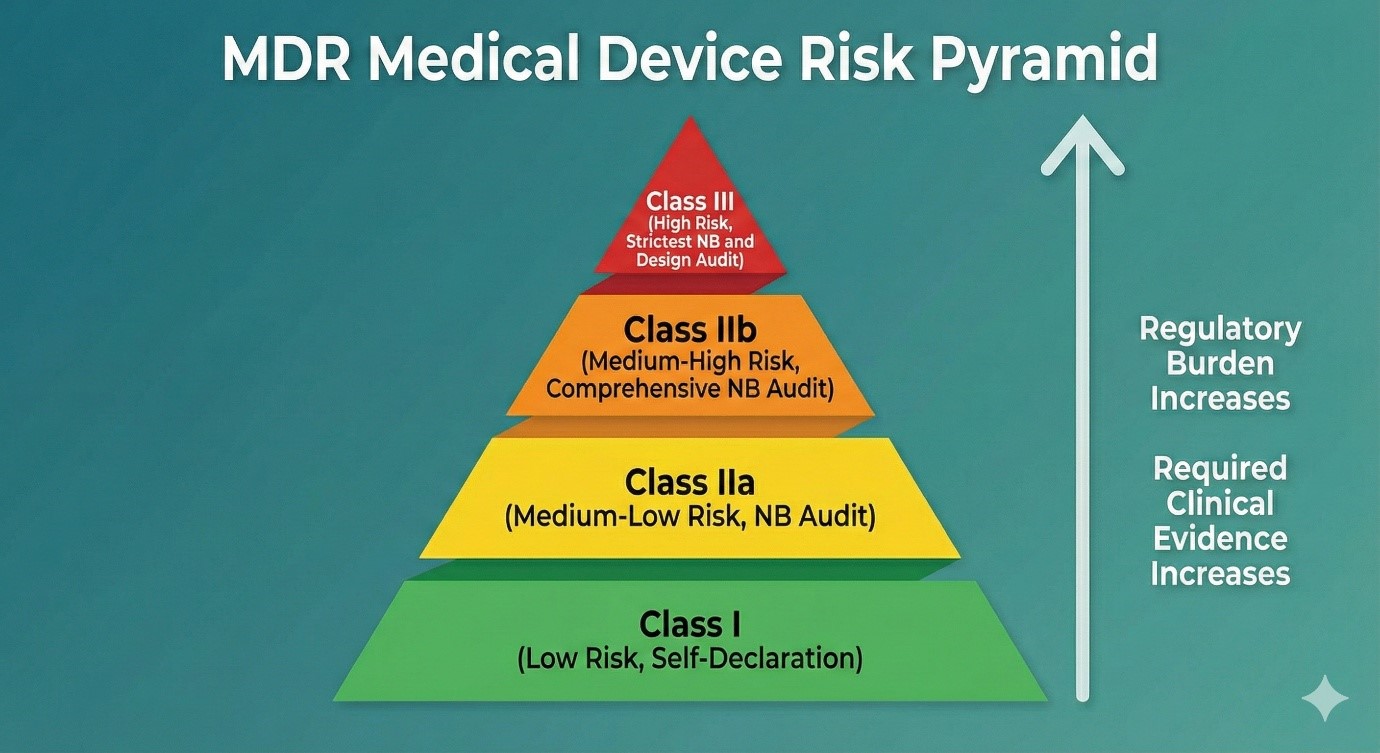
Section 1: What is a Medical Device? Definition and Scope
A New Era in Biosimilar Development: FDA Reform and Implications for Clinical Research Centers
Prof. Dr. Işık Tuğlular honored our Phase 1 Clinical Research Center
Our Clinical Research Center’s Website is Now Live!
We are pleased to announce the official launch of our Clinical Research Center’s website for our valued visitors and partners.
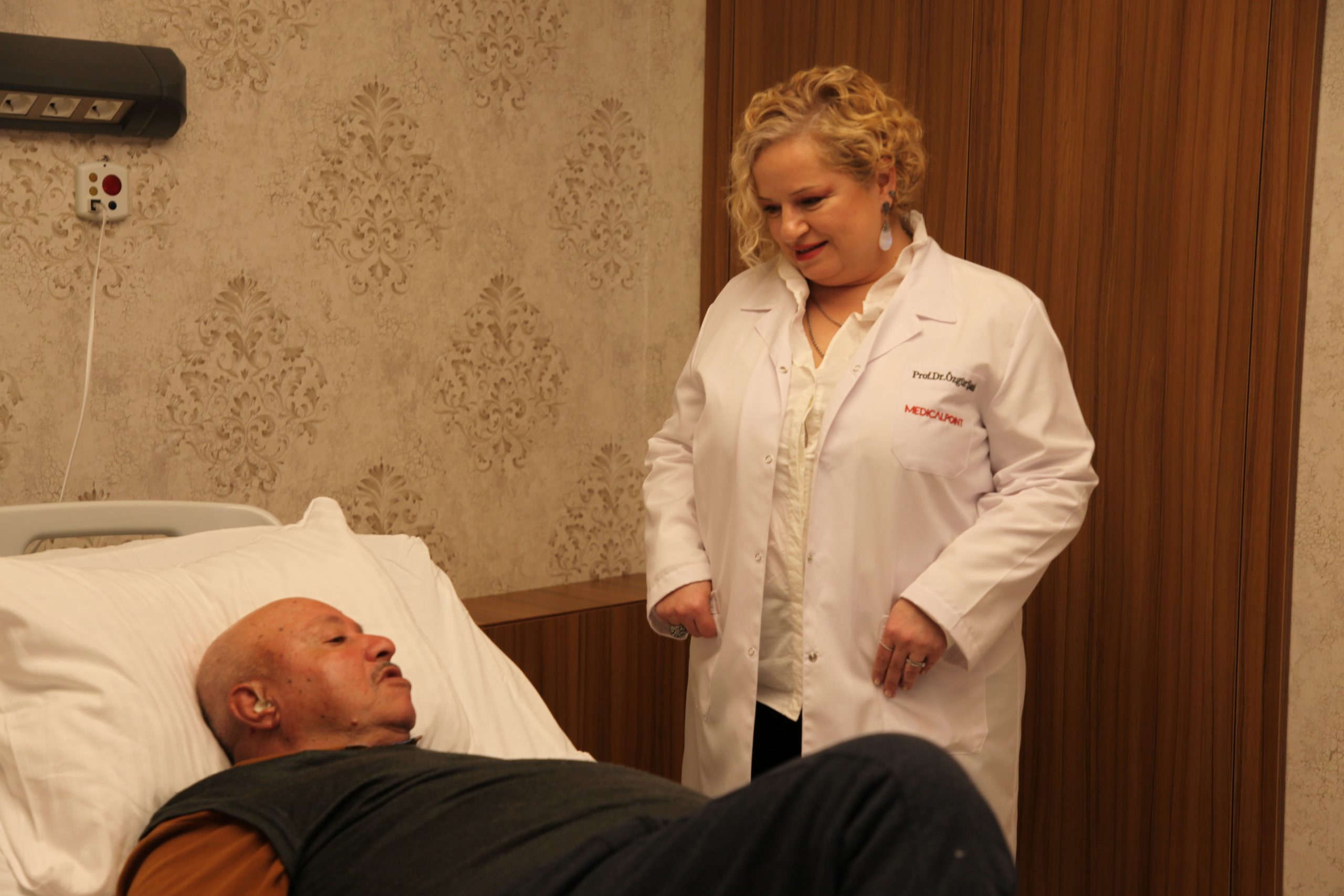
Promising Treatment with Lutetium
Prof. Dr. Özgür Şanlı from the Nuclear Medicine Department of İzmir University of Economics (IEU) Medical Point Hospital discussed the importance of “Lutetium” therapy for prostate cancer patients who have not benefited from treatments like chemotherapy and radiotherapy, stating, “It is possible to overcome prostate cancer with Lutetium treatment.”
IEU Clinical Research Ethics Committee Begins Its Work Under the New Regulation
The İzmir University of Economics Clinical Research Ethics Committee, established under the “Regulation on Clinical Trials of Human Medicinal Products” dated 27.05.2023 and approved by TİTCK on 24.07.2024, has commenced its activities.
Visit from Harvard Doctors to Medical Point
Prof. Dr. Gökhan Hotamışlıgil, Director of the Sabri Ülker Center for Metabolic Research at Harvard University, and his team visited İzmir University of Economics Medical Point Hospital to discuss potential collaboration on clinical research.
Clinical Research Center Opens at Medical Point
The Medical Point Gaziantep Hospital is proud to be the first private hospital in the Southeastern Anatolia Region to establish a Clinical Research Center.