Abstract: By 2026, the world of clinical research will have transformed into a multidimensional technology ecosystem where data science, artificial intelligence, and sophisticated regulatory strategies are intertwined (1). This article examines synthetic control arms, artificial intelligence-based protocol automation, and global regulatory updates in 2026 from an academic perspective (2).
- Introduction
Clinical research is undergoing a historic turning point in the processes of generating medical knowledge and testing the reliability of new treatments (3). While traditional randomised controlled trials (RCTs) remain the gold standard, cost constraints, ethical concerns, and patient participation challenges have driven the sector towards innovative models such as digital twins and synthetic control arms (4).
- Methodological Innovations: Synthetic Control Arms (SCA)
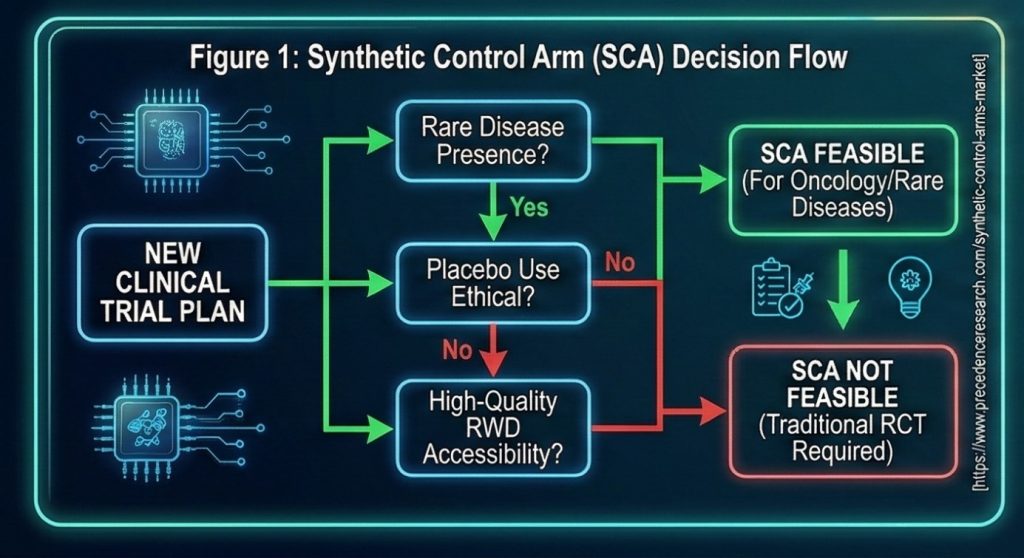
Synthetic Control Arms (SCA) involve creating a virtual comparator group using real-world data (RWD) or historical clinical trial data, without enrolling new patients in a study (4). As of 2026, the clinical successes and market data for this method are as follows:
- Ethical Advantage: Particularly in oncology and rare diseases, it eliminates the risk of patients being assigned to the placebo group, ensuring that every participant has access to active treatment (5).
- Data Sharing and Success: Comprehensive oncology studies presented in 2025 demonstrated that synthetic data sets created with artificial intelligence showed complete consistency with original data in survival analyses (6).
- Market Distribution: According to 2026 projections, real-world data (RWD) will maintain its leadership in SCA technologies with 53.50%, while AI-based analysis platforms will hold a 37.50% market share (4).
- Artificial Intelligence and Protocol Automation
Artificial intelligence has become a cornerstone of clinical operations by 2026 (7). Its impact is particularly tangible in protocol design and data management:
- Increased Efficiency: AI-supported electronic clinical outcome assessment (eCOA) workflows have reduced data errors by 74% and shortened review cycles by 67% (2).
- Digitalisation: The automatic conversion of PDF protocols into electronic data capture (EDC) systems and the use of artificial intelligence to write new trial protocols based on historical data (digitalisation) minimises set-up times (7).
- Regulatory Transformation: FDA, EMA and TİTCK Updates
Technological advances have prompted regulatory authorities to adopt new policies focused on ‘evidence optimisation’ (8):
- FDA (19 February 2026): The FDA has officially confirmed that a single pivotal study may be sufficient for approval under certain conditions (8). This necessitates a ‘total evidence ecosystem’ approach supported by biomarker alignment and real-world evidence (8). Additionally, a ‘Plausible Mechanism’ framework has been published for ultra-rare diseases (9).
- EMA (15 January 2026): The European Medicines Agency has implemented a new Variation Guidance to facilitate the digitalisation and streamlining of post-marketing processes (10). The ICH M14 guideline, which came into force in March 2026, has introduced international standards for the use of observational data in safety assessments (11).
- TİTCK (14 November 2025): In Turkey, it has become mandatory to submit an internationally recognised Good Laboratory Practice (GLP) certificate for clinical trial applications (12).
- Decentralised Clinical Trials (DCT)
Hybrid and decentralised models have become standard practice by 2026 thanks to wearable technologies (2). Wearable devices continuously monitor over 200 parameters, such as sleep and movement, reducing the burden on patients to visit the centre and improving data quality (2).

- Conclusion
Clinical research is evolving into a faster, more ethical, and patient-centred structure with the analytical power of artificial intelligence (13). The artificial intelligence market, expected to reach $190 billion by 2030, demonstrates that this transformation is permanent and strategic (1).
- Gündüz Y. Haberler. hospitalmanager [İnternet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.hospitalmanager.com.tr/haberler/
- MBiotech SS. Top Clinical Trial Trends for 2026: Decentralization, AI Recruitment and RWD. Xtalks [Internet]. 24 Kasım 2025 [a.yer 04 Mart 2026]. Erişim adresi: https://xtalks.com/top-clinical-trial-trends-for-2026-decentralization-ai-recruitment-and-rwd-4502/
- Clinical Research in 2026:What’s Changing and Why It Matters [Internet]. 22 Ocak 2026 [a.yer 04 Mart 2026]. Erişim adresi: https://cromospharma.com/clinical-research-in-2026-what-changed-and-why-it-matters/
- Synthetic Control Arms Market Size, Report by 2034 [Internet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.precedenceresearch.com/synthetic-control-arms-market
- nashvillebios. Synthetic Control Arms in Clinical Trials. Nashville Biosciences [Internet]. 18 Aralık 2024 [a.yer 04 Mart 2026]. Erişim adresi: https://nashbio.com/blog/clinical-trials/synthetic-control-arms-in-clinical-trials/
- How AI is expediting clinical research: the use of synthetic real-world data [Internet]. [a.yer 04 Mart 2026]. Erişim adresi: https://dailyreporter.esmo.org/esmo-ai-digital-oncology-congress-2025/editorial/how-ai-is-expediting-clinical-research-the-use-of-synthetic-real-world-data
- Trends for 2026: Targeted AI, continuous trials, and navigating uncertainty [İnternet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.merative.com/blog/clinical-trial-trends-2026
- FDA’s Shift to One Pivotal Trial for Drug Approval: Strategic Implications for Sponsors [Internet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.propharmagroup.com/thought-leadership/fda-default-drug-approval-requirements-one-pivotal-trial-regulatory-strategy-implications
- Editor AS Senior. FDA Introduces New Framework to Advance Individualized Therapies for Ultra-Rare Diseases | Applied Clinical Trials Online [Internet]. 2026 [a.yer 04 Mart 2026]. Erişim adresi: https://www.appliedclinicaltrialsonline.com/view/fda-new-framework-individualized-therapies-ultra-rare-diseases
- Guidance on the application of the revised variations framework | European Medicines Agency (EMA) [Internet]. 2024 [a.yer 04 Mart 2026]. Erişim adresi: https://www.ema.europa.eu/en/guidance-application-revised-variations-framework
- Regulatory Intelligence – 2025 Round Up — Scendea [İnternet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.scendea.com/articles/regulatory-intelligence-2025-round-up
- TİTCK Tarafından Klinik Araştırma Başvurularında Güven Uygulamaları Kılavuzu Güncellenmiştir – Duyurular [İnternet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.zumbul.av.tr/tr/duyurular/t-tck-taraf-ndan-klinik-ara-t-rma-ba-vurular-nda-guven-uygulamalar-k-lavuzu-guncellenmi-tir
- Team F. Trends in Clinical Trials in 2025: Navigating a Transformative Landscape. Florence [Internet]. [a.yer 04 Mart 2026]. Erişim adresi: https://www.florencehc.com/blog-post/trends-in-clinical-trials-in-2025-navigating-a-transformative-landscape/


